
نشریه علمی پژوهشی طب انتظامی Journal of Police Medicine

Volume 12, Issue 1 (2023)
J Police Med 2023, 12(1) |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Fatollahi Arani S, Zeinoddini M. Gene editing: biosecurity challenges and risks. J Police Med 2023; 12 (1)
URL: http://jpmed.ir/article-1-1171-en.html
URL: http://jpmed.ir/article-1-1171-en.html
1- Department of Biology, Faculty of Chemistry & Chemical Engineering, Malek Ashtar University of Technology, Iran
2- Department of Biology, Faculty of Chemistry & Chemical Engineering, Malek Ashtar University of Technology, Iran ,zeinoddini@modares.ac.ir
2- Department of Biology, Faculty of Chemistry & Chemical Engineering, Malek Ashtar University of Technology, Iran ,
English Extended Abstract: (6885 Views)
INTRODUCTION and HISTORY of GENE EDITING
After the first presentation of the term biotechnology by Karl Erky, a Hungarian agricultural engineer, perhaps few people imagined this technology would be used in offensive aspects and against human society. At the beginning of the work, biotechnology was presented as a clear and suitable solution for human society to create suitable treatment conditions, healthy nutrition, better life and a hopeful future. However, over time, the dark aspects of this technology in the form of bioterrorist threats were brought up, and this led to the use of the title "Dark Biotechnology" for bioterrorism attacks in the colored names of biotechnology. [1, 2]. In the military developments of the last century, which were rooted in technology, various scientific branches, including modern chemistry and physics, have been the main factor. Current trends indicate that the next evolution will be rooted in biological science. The development of biological technology has facilitated the development of biological weapons and threats, and the third wave of technology in the history of the development of weapons of mass destruction will be biological. The possibility of dual military and civilian use lies in biotechnology. In other words, biotechnology can be helpful and harmful. Based on this, the sciences related to biology, especially genetic engineering and biotechnology, in addition to being able to be used to advance medical and therapeutic sciences, at the same time, these researches can be developed with the cover of medical research in the military field and every day designed and produced newer biological agents. In the first case, we will see progress in human health and society, but in the second case, it will cause bioterrorism attacks and human deaths. Such threats result from new technologies that, in addition to making progress in science and technology, also enable the production of new microorganisms (artificial synthesis) [3-6]. It is necessary to explain that in 2012, an American person published an article entitled "The Future of Biological Threats" in the Journal of Microbial Biotechnology, in which he claims one of the three theories of the extinction of human society, after the possibility of a large-scale nuclear war and a vast meteorite collision to the ground, causing contagious infectious disease [7]. The turning point of developments related to biotechnology was the beginning of the human genome project, which began in 1991, and finally, with the holding of an international conference in the White House (2000), the completion of the human genome project to the global community with the presentation of the main executives of this project (Francis Collins and Craig Venter) and was announced with the presence of the President of the United States (Clinton). With the completion of the human genome project, complete information about the human genome was provided to human society. With this information, the defective and functional genes were fully identified. Therefore, the sensitivity of individuals and families to microorganisms and dangerous diseases or their resistance was determined [8, 9].
Gene therapy has historically been defined as adding new genes to human cells to treat genetic diseases. However, the recent advent of genome editing technologies has created a new paradigm in which the human genome sequence can be precisely manipulated to achieve a therapeutic effect; this involves correcting disease-causing mutations, adding beneficial genes to specific locations in the genome, and removing harmful genes or genome sequences. Understanding the genetic basis of hereditary disease led to the initial concept of gene therapy, in which suitable foreign DNA replaces defective DNA in people suffering from genetic defects. More than 40 years of research in the field of gene therapy process shows that the simple idea of gene replacement is much more complicated to perform safely and effectively [10, 11]. Many of these challenges have focused on fundamental limitations in precisely controlling how genetic material is transferred into cells. Nevertheless, there are technologies for adding foreign genes that have made significant progress in this field. Potential clinical results have now been demonstrated in a wide range of strategies and medical indications, but several challenges remain. Integrating therapeutic transfer genes into the genome to maintain stability in the cell may affect gene expression and its unwanted effects on nearby genes. In addition, some genes need to be more significant to transfer by vectors quickly. Finally, foreign genes cannot always be introduced directly into dominant mutations or defective genetic material. To solve the problems related to these basic limitations, conventional methods have emerged to make precise and targeted changes in the genome [12, 13]. In this regard, genome editing is a practical, versatile, and preferred tool for functional gene research, gene therapies, and precise breeding of crops and domestic and attractive animals for practical and industrial research [14, 15].
Genome editing research started in the 1970s. The first major step in gene editing was achieved when researchers showed that when a piece of DNA enters a cell, it can enter the host genome through homologous recombination and implement the desired changes in the cell. This development came when it was found that in eukaryotic cells, more precise gene targeting mechanisms could be achieved by inducing a double-strand break in a specific genomic target. In addition, the scientists found that if a synthetic DNA restriction enzyme was introduced into the cell, it could recognize the DNA at specific locations and cut it into double strands, subsequently repaired by HDR (Homology-Directed Repair) and NHEJ (Non-Homologous End-Joining) mechanisms resulting in homology-based insertions, deletions, or repairs [16, 17]. Among the different methods and mechanisms of gene editing, CRISPR-Cas9 technology (the fourth generation of gene editing) has surpassed other methods. Two female scientists introduced this technology to the scientific community in 2012, making them proud to receive the Nobel Prize in Chemistry in 2020. It is necessary to explain that some sources have announced the beginning of the first research related to gene editing in 1987, which indicates the existence of initial ideas in this regard. However, its expansion has been observed since 2000. CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeat, which has been mentioned in various sources. The CRISPR-Cas9 system was developed after other gene editing systems called meganucleases (a class of endodeoxyribonucleases), ZFNs (Zinc-Finger Nucleases) and TALENs (Transcription Activator-Like Effector Nucleases) due to its unique features, it attracted the attention of researchers, so the publication of articles and inventions in this field shows great growth (Figure 1). It should be noted that most of the research in this regard is dedicated to cancer, AIDS, and hepatitis [18, 19].
MECHANISMS of GENE EDITING
Gene editing can change an organism's DNA sequence, essentially engineering its genetic makeup. This process is carried out using enzymes, specifically nucleases that are engineered to target a specific DNA sequence. They introduce cuts into the DNA strands, allowing the removal of existing DNA and inserting replacement DNA. In other words, the gene editing tools developed today can create double-stranded breaks in the genome, and by repairing these breaks, the process of gene editing can be developed. There are four methods of gene editing: gene destruction or mutation, gene deletion, gene modification and gene insertion. Based on this, researchers use different tools capable of creating double-strand breaks in DNA to create various changes in the genome. Specific nucleases for gene editing include engineered target sequences and restriction enzymes. After the programmed nuclease cleaves the target gene to introduce double-strand breaks (DSBs), molecular repair proceeds via two fundamentally different mechanisms: homology-directed repair (HDR), in which broken DNA is repaired using a homologous DNA sequence as a template, and non-homologous end joining (NHEJ), in which broken ends in a non-homologous DNA sequence are rejoined. The HDR repair mechanism, which allows the insertion of a template DNA to correct or insert a particular sequence at the site of a DNA break, facilitates accurate copying of the template to a specific location in the genome and repairs the homologous DNA break.
In contrast, the NHEJ repair mechanism leads to small insertions or deletions (indels) at the desired site or breaks [20, 21]. As a result, this mechanism can be an efficient way for defective genes to function. As mentioned, today, four different types of nucleases that bind to DNA are used in gene editing: Meganucleases, Zinc-Finger Nucleases (ZFN), Transcription Activator-Like Effector Nucleases (TALEN) and Cas9 Nuclease, which is the most recently discovered type [22, 23]. Table 1 compares these four nucleases regarding features and mechanism of action. Their performance is also shown in Figure 2.
ADVANTAGES of GENE EDITING TECHNOLOGY
Gene editing is essential and valuable in various industrial and research fields. In the continuation of the recent exciting developments in the ease of use, features and characteristics of gene editing technology and their application in different fields are examined [24-26].
Human Health: Gene editing technology creates a fundamental change in gene therapy. It can treat a wide range of diseases (such as diabetes, cancer, cystic fibrosis and sickle cell anaemia) that have not been possible to treat so far using this technology. All cancers result from numerous diverse mutations that lead to the excessive growth and proliferation of cells and the emergence of malignant phenotypes. The site of the event and the area disrupted by these mutations can be classified into four categories: oncogenes, tumor suppressors, epigenetic factors and chemotherapy resistance genes. CRISPR-Cas9 technology, as a powerful tool with high characteristics, can correct these mutations and treat cancers derived from them. Since oncogenic changes in several cancers lead to increased cell proliferation and malignancy, oncogenes such as tyrosine kinase receptor Erb2 can be directly targeted by CRISPR-Cas9 technology. From a complementary point of view, the CRISPR-Cas9 method can create cancer-causing mutations in human cell lines and animal models. In this regard, lung cancer cell lines, acute myeloid leukaemia, and liver and pancreatic cancer have been developed. Crisper-Cas9 technology can also be used in animal models suffering from various diseases (from hereditary diseases to cancers). It has created heritable changes by CRISPR-Cas9 technology and direct targeting of one or more bilateral in the animal egg. Among the transgenic animal models, most of the tests are based on mouse models. However, researchers have succeeded in making models of non-human primates by targeting multiple genes. The advantage of these models is in the reconstruction and the possibility of investigating complex human diseases such as neurodegenerative diseases. However, mouse models have more advantages than others, such as the cost of working with them, and in addition, mouse models are very suitable for extensive mutagenesis studies in vivo [25].
New Materials: By using these technologies, it is possible to achieve the synthesis of new materials that can be used in various applications, such as the release of oral drugs or the production of biosensors.
Drug Development: Engineered cells can be produced using these technologies that produce drug production optimally and efficiently. In addition, it significantly reduces the cost of the drug and provides easy access to the drug.
Research Applications: With technologies such as gene editing, new animals and cell models can be designed and produced, which will help us learn more about diseases and test new drugs and vaccines on those cell and animal models.
Agriculture: By using gene editing tools, it is possible to modify the seeds of agricultural products without harming other genes. Based on this, it is possible to obtain agricultural products that can be resistant to infections and environmental damage, and as a result, food security can be improved.
Bioenergy: With the help of tools such as gene editing, it is possible to produce biofuels (green). Therefore, it is possible to increase and optimize the production of biofuels such as ethanol in algae cells or seeds by modifying the metabolic and biochemical pathways of the relevant cells [24-26].
Criminology: By combining CRISPR technology with DNA fingerprinting technology, new methods can be developed in criminal identity detection and criminology. DNA (genetic) fingerprinting is a method that was first presented to the scientific community and criminology experts in 1985 by Alec Jeffrey using repeatable variable sequences of 15 to 100 games called VNTR (Variable Number of Tandem Repeat). Genetic fingerprinting allows specialists to determine the differences and similarities between people based on specific DNA samples. Based on this, people's communication can be observed and identified at crime scenes. According to each person's VNTR sequence, gRNA (gene editing diagnostic tool) can be designed. Based on this, a fluorescently labelled DNA sample from the victim (in a crime) can be prepared and checked for matching with the VNTR sequence of the victim. CRISPR can also be designed to scan DNA or find a specific VNTR. In the CRISPR DNA scan, if CRISPR fails to target the VNTR, it will not bind to it, meaning no fluorescent dye will appear under UV light. However, if scanning is performed and the target is identified and bound, a fluorescence signal is generated, meaning the VNTR can be present in the DNA [27].
RISKS of GENE EDITING TECHNOLOGY
In today's world, the increasing importance of the knowledge of biology as a basic science is undeniable. As a result of deep studies and many investigations, the boundaries of biology and the findings related to the knowledge of nature have been expanded tremendously. The volume of resulting information and its increasing growth cannot be compared to any era. Today, biotechnology, as a branch of biological applications, has progressed more than at any other time. Due to its applications in health, hygiene and the economy, its importance and value have increased daily. These important advances in biotechnology are mainly due to advances in instrumentation and their application in developing the frontiers of biology. The most remarkable developments of this knowledge and technology have been achieved in ecology, genetics, microbiology, molecular biology, biochemistry, cell culture technologies and process engineering. The emergence of the new sciences of genomics, proteomics, bioinformatics, systems biology, synthetic biology and gene editing has also resulted from these developments. On the other hand, today, biology is exposed to hostile abuse as much as chemistry in World War I and physics in World War II. The enormous force of international trade that underpins this basic science has driven it to innovations that, along with its marketable medical value, may also be used for destructive purposes. If a country exploits science and technology in biological fields, it can reveal one of the most serious problems of humanity that it has not faced so far. If the production of a new generation of bioweapons is pursued with force, especially if used to control and conquer humans, it can cause dangerous technological competition. If the force of biotechnology is not politically contained, it will be able to invent scientific methods that will change the way war is conducted and increase the means of civilian sacrifice. It should be noted that the discussions discussed below do not in any way attempt to magnify the possible risks of biotechnology; because such a possibility exists with any other technology, both those that are widely used in societies today (such as IT and telecommunications) and those that will emerge in the future in the field of health (such as gene editing, artificial synthesis, and human creation). Such studies can also show the potential capabilities of biotechnology in promoting new defense capabilities; so that the guardians of the country's biological defense pay special attention to the potential of this technology for defense purposes (such as providing new treatment trends and new diagnostic methods in the direction of biological defense).
Among the various technologies proposed in the field of life and health today, gene editing technology can cause irreparable risks to the biological security of societies. Now genome editing is much easier, faster, cheaper and more efficient than ever before (just like editing an article on a computer) and helps researchers in various scientific fields. The fourth generation of gene editing, CRISPR-Cas9, can handle new editing programs, from viruses and bacteria to animals, plants and humans. However, as this technology develops, how should it be controlled? While expressing concern about the negative consequences of the development of gene editing technology, Professor Jennifer Doudna, the discoverer of Cas9 nuclease and 2020 Nobel Prize winner, has received $3.3 million in funding from the US Defense Advanced Research Projects Agency (DARPA) to investigate anti-CRISPR solutions. The red line of this technology is the manipulation of the human embryo [28-30].
It should be remembered that CRISPR-based genome editing technology has caused a tremendous revolution in medical sciences and other scientific fields. This technology, developed for about a decade, has fascinated scientists. In a way that it allows a person with less than a high school education to edit the genome of any animal or plant. High school students are now using this technology to perform experiments that were previously only dreams for most scientists. Much of the scientists' research has focused on the tremendous potential of using genome editing to treat cancer. This work is based on the familiarity and understanding of the new model of the cancer network, which examines how cancer cells are controlled. New gene-based approaches for cancer treatment have suggested using genome editing as an effective tool. Unfortunately, while we can cure cancer using CRISPR, we can also create cancer using the same technology.
Based on this, researchers will have been able to design cancer models on the computer. The possibility of artificially creating cancerous tumors will also be provided when they can treat cancer patients with CRISPR. Therefore, the cancer bomb can be implemented using CRISPR. As a result, today, CRISPR technology is proposed as a new biological weapon, and scientists warn about its biosecurity consequences [31, 32]. The very simple application of CRISPR makes this technology potentially very dangerous. This technology has features that make it an ideal military and bioterrorist weapon and has been favored by biohackers. Mr. Josiah Zayner, one of the pioneers of biohacking and the founder of Odin Company since 2006, offers online training and simple gene therapy kits based on CRISPR technology to his customers around the world, who, in addition to the simple training of gene editing, can design and produce genetically engineered products (such as the green tree frog with the high growth rate) [33].
Based on this, it seems that in the not-too-distant future, we will see the design and production of biological weapons (mass destruction) based on CRISPR, which will be even more dangerous than a nuclear bomb. According to some scientists, nuclear weapons are obsolete because they are complicated to maintain. From a military point of view, CRISPR weapons are considered far superior to nuclear weapons and will probably replace them. These CRISPR-based editing features make it very easy to weaponize. It is possible to create an engineered virus that edits CRISPR in a completely controlled manner so that only humans whose genomes have specific characteristics are killed or disabled by the virus. The importance of CRISPR biosecurity is so high that scientists have warned about the design and production of programmed humans and editing on human embryos and are drafting national and international laws in this regard. It is necessary to explain that the Chinese scientist, He Jiankui, on October 8, 2018, officially announced the birth of Chinese girl twins with Crisper technology. In these twins, who were born from a father with AIDS and a healthy mother, the CCR5 gene, which is related to the entry of HIV into cells, has been edited and deleted. This scientific activity, carried out illegally and confidentially, shocked the scientists in this field and led to a fine and three years of imprisonment for this Chinese scientist. Jiankui had to present the result of his research at the Second International Conference on Human Genome Editing held in Hong Kong in 2018, which was widely criticized by the scientific community, even Chinese scientists [34].
The question raised today is why the Crisper weapon can be so dangerous. The answer to this question can be found in the following:
1- CRISPR-Cas based editing can be precisely designed to edit a specific part of the target genome.
2- Viruses can deliver CRISPR-Cas-based editing to a given host.
3- Mathematical rules can fully control edits. In other words, target genome editing is applied only if certain precise conditions are present in the target person's genome. For example, two people can be infected by a CRISPR-based editing virus, but only the person with the prerequisites will have their genome edited. In addition to these, unfortunately, many more characteristics of a potential CRISPR-based weapon make them ideal weapons for future precision and targeted mass destruction. For example, its effects can remain hidden for months, and CRISPR bombs do not have the long-term toxic effects of nuclear weapons. Also, it can be implemented for most organisms and programmed as an effective weapon for the nervous system. As a result, CRISPR as a weapon of mass destruction for genocide is significant and considered [34, 35]. Also, from a biosecurity perspective, CRISPR technology could potentially create and induce precise cancers that would kill people within months. Unfortunately, it is much easier to cause cancer than to cure it with genome editing. The main appeal of a deadly CRISPR-engineered virus as a bioterrorism weapon is the precision in mass incapacitation and destruction of people. Unfortunately, these topics are unrelated to science fiction and fantasy horror movies, but this is a real danger in humanity's present and future. Notably, there are probably labs worldwide developing CRISPR technology to develop the next generation of bioweapons. As mentioned, Crisper also has genocidal capabilities. Given that a particular generation has unique genetic traits that distinguish it from others, all members are potential targets for a CRISPR-engineered killer virus. For example, if the prerequisite is that one must have brown eyes, then anyone with brown eyes is a potential target for a deadly CRISPR-engineered virus. Using the Crisper weapon, you can create diseases that cause the target person to die slowly or quickly. Some scientists who have provided significant analysis about the possible possibilities have raised various dangerous issues. Some of these reports indicate major future concerns that could result from secret research programs. Especially these reports have not ruled out the possibility of hidden viruses that can secretly enter the genome of a population and later be activated by a signal. Another example is "programmed cell death". This ability to insert a genome into the genetic reserve of a specific population and attack it at will, or to create a completely new pathogenic agent, indicates a change in capabilities [35, 36].
Gene Editing Defence Strategies
On June 29, 2018, in Newsweek magazine, the media announced and published the discussion of using gene editing technology on human embryos to eliminate genetic diseases. However, some scientists and pioneers of this technology (such as Professor Doudna) raise ethical considerations as a serious discussion. Since 2015, China has started extensive research on human embryos under the leadership of Professor Lu You, an oncologist at Sichuan University in Chengdu, and is still developing this technology on human embryos; in a way that on October 28, 2017, a group led by him injected the modified cells into a patient with aggressive lung cancer as part of a clinical trial at the West China Hospital [37]. However, according to most scientists, the red line of gene editing technology is the genetic manipulation of human embryos. However, today, some scientists secretly use gene editing technology to research and develop human embryos to design and create disease-free humans with special capabilities. These designed children have high intelligence, creative mind, five senses with great power and are resistant to various diseases. Also, some have called this technology the engine of creation because it gives the scientist a god-like power (based on his beliefs) to create and improve future humans (super-humanity). On the other hand, unlike the Schilling Act, CRISPR-based bioweapons have international strategic implications. According to Thomas Schelling's Game Theory, a balanced international strategy is observed in the nuclear age. If one side increases or decreases its nuclear weapons capabilities to maintain balance, the other side must follow suit. Based on this, each side knows what weapons the other side has, and both sides, knowing this (based on open inspection of nuclear facilities), observe a balance in this regard. However, in the era of genome editing technology, due to the ease of making CRISPR-based biological weapons in small laboratories that are impossible to observe and identify, the awareness of the ability and capacity to design and manufacture biological weapons on the other side has failed and as a result, the strategy based on inspection and mutual destruction of such weapons of mass destruction also fails. Therefore, CRISPR-based bioweapons represent a fundamental change in Schilling's Law. A balanced, no-win, bargaining, nonzero-sum Schilling game approaches the classical zero-sum game, in a way that the side that attacks first may win the entire game. As a result, according to Schelling's argument, this international situation will be very unstable, fragile and dangerous [35].
Based on this, how can the defense strategy to deal with the threats resulting from such technologies and biological security be? In other words, how do we defend ourselves against biological weapons caused by CRISPR? To answer these questions, it is important to pay attention to the following:
1- The attention and awareness of public opinion and political leaders should be increased about the dangers of CRISPR-based genome editing.
2- Considering the actual and potential risks of CRISPR-based genome editing in bioterrorist attacks, defense strategies should be quickly developed to deal with such possible attacks.
3- Any CRISPR-based editing can be reversed. In other words, a network mutation that causes cancer can be reversed to stop cancer. When we know how to stop cancer, we will know how to cause cancer. Alternatively, any gene essential for life can be disabled by a reverse edit. This requires a coherent defense-research initiative.
4- It is necessary to adopt coherent international laws (in line with the Convention on the Prohibition of the Development, Production and Stockpiling of Biological Weapons) to examine the risks of this very dangerous technology (in order to confront and not prevent it).
5- A new international bioethics committee is needed to deal with risk groups in this regard.
Conclusion
Gene editing technology has shown the scientific community a new window and solution through which appropriate treatment methods can be adopted to deal with most diseases. Therefore, genome editing offers great opportunities in biology, biotechnology and medical sciences, including preventing and treating diseases and producing good food. On the other hand, CRISPR-based bioweapons destroy the logical and strategic balance of power that has kept the world free of catastrophic wars. The world is facing a potentially more dangerous technology than nuclear weapons because of its ease of development and precision of use. Precise targeting of individuals with a deadly CRISPR-based virus means that, as in nuclear war, there is no longer a barrier to mutually assured destruction. Instead, we are faced with the possibility of a precise and targeted mass genocide. On the other hand, after the birth of the Chinese gene-edited twins, in terms of Islamic jurisprudence and ethics, there have been discussions about the possibility of using CRISPR technology to develop embryonic research and human genome editing. Considering that embryonic gene editing may lead to hereditary changes in the human genome, whether this practice should be permissible requires a deep and detailed discussion from different perspectives. Islam's views on the concerns raised about human genome editing consider the moral principles important in Islam and declare that it should be taken into account when evaluating the permissibility of gene editing of the human reproductive line through CRISPR. As discussed in this article, human embryo editing research for medical purposes is legal under certain conditions and is used to treat diseases, but until the safety and effectiveness issues of this technology are resolved, it should not be applied to humans. Strong and strict ethical guidelines are necessary to preserve human dignity and prevent the misuse of technology, and religious principles of preserving human life, descent and dignity and preventing possible harm are among the important principles in evaluating the permissibility of human embryo editing through CRISPR from an Islamic point of view. Therefore, it can be concluded that human gene editing by CRISPR is considered halal in Islam if it has the following conditions:
A- To be used only for medical purposes, especially for preventing or treating diseases. This kind of change is not considered a manipulation of God's creation.
B- It is allowed only after solving the safety and efficiency problems, and the technology used should not cause more harm to the parents, the resulting child, the society and the future generation.
C- Establish strict regulations to ensure respect for the people involved, prevent early use and abuse of technology, and seriously prevent unwanted genetic changes in humans [38-40].
Clinical & Practical Tips in POLICE MEDICINE: Considering the emergence of promising technologies such as genome editing, in addition to using the opportunities obtained from this technology, especially in the field of life and health and diagnosis and treatment of incurable diseases, it is necessary to pay special attention to this technology from the point of view of biosecurity. The creation of new organisms, biohacking and the creation of programmed humans are among the future biosecurity risks of this technology, and the Deputy Health and Medical Services of General Staff of the Armed Forces must draw specific plans and strategies in this regard in cooperation with academic centers.
Acknowledgements: This study was carried out in the Research Institute of Biological Sciences and Technology of the Malek Ashtar University of Technology, and the efforts of the officials of this university are gratefully acknowledged.
Conflict of interest: The article's authors stated that the present study has no conflict of interest.
Authors' Contribution: Samane Fethollahi (data collection); Dr Mehdi Zein al-Dini (presenting the idea and design of the study, data collection, data analysis). All the authors participated in the initial writing of the article and its revision, and all accept the responsibility for the accuracy and correctness of the article's contents with the final approval of this article.
Financial Sources: The present study was carried out with the financial support of the Biological Sciences Department of the Academic Complex of Chemistry and Chemical Engineering of Malek Ashtar University of Technology, in line with the observation of emerging technologies, and was a part of the research project number "19730180208".
After the first presentation of the term biotechnology by Karl Erky, a Hungarian agricultural engineer, perhaps few people imagined this technology would be used in offensive aspects and against human society. At the beginning of the work, biotechnology was presented as a clear and suitable solution for human society to create suitable treatment conditions, healthy nutrition, better life and a hopeful future. However, over time, the dark aspects of this technology in the form of bioterrorist threats were brought up, and this led to the use of the title "Dark Biotechnology" for bioterrorism attacks in the colored names of biotechnology. [1, 2]. In the military developments of the last century, which were rooted in technology, various scientific branches, including modern chemistry and physics, have been the main factor. Current trends indicate that the next evolution will be rooted in biological science. The development of biological technology has facilitated the development of biological weapons and threats, and the third wave of technology in the history of the development of weapons of mass destruction will be biological. The possibility of dual military and civilian use lies in biotechnology. In other words, biotechnology can be helpful and harmful. Based on this, the sciences related to biology, especially genetic engineering and biotechnology, in addition to being able to be used to advance medical and therapeutic sciences, at the same time, these researches can be developed with the cover of medical research in the military field and every day designed and produced newer biological agents. In the first case, we will see progress in human health and society, but in the second case, it will cause bioterrorism attacks and human deaths. Such threats result from new technologies that, in addition to making progress in science and technology, also enable the production of new microorganisms (artificial synthesis) [3-6]. It is necessary to explain that in 2012, an American person published an article entitled "The Future of Biological Threats" in the Journal of Microbial Biotechnology, in which he claims one of the three theories of the extinction of human society, after the possibility of a large-scale nuclear war and a vast meteorite collision to the ground, causing contagious infectious disease [7]. The turning point of developments related to biotechnology was the beginning of the human genome project, which began in 1991, and finally, with the holding of an international conference in the White House (2000), the completion of the human genome project to the global community with the presentation of the main executives of this project (Francis Collins and Craig Venter) and was announced with the presence of the President of the United States (Clinton). With the completion of the human genome project, complete information about the human genome was provided to human society. With this information, the defective and functional genes were fully identified. Therefore, the sensitivity of individuals and families to microorganisms and dangerous diseases or their resistance was determined [8, 9].
Gene therapy has historically been defined as adding new genes to human cells to treat genetic diseases. However, the recent advent of genome editing technologies has created a new paradigm in which the human genome sequence can be precisely manipulated to achieve a therapeutic effect; this involves correcting disease-causing mutations, adding beneficial genes to specific locations in the genome, and removing harmful genes or genome sequences. Understanding the genetic basis of hereditary disease led to the initial concept of gene therapy, in which suitable foreign DNA replaces defective DNA in people suffering from genetic defects. More than 40 years of research in the field of gene therapy process shows that the simple idea of gene replacement is much more complicated to perform safely and effectively [10, 11]. Many of these challenges have focused on fundamental limitations in precisely controlling how genetic material is transferred into cells. Nevertheless, there are technologies for adding foreign genes that have made significant progress in this field. Potential clinical results have now been demonstrated in a wide range of strategies and medical indications, but several challenges remain. Integrating therapeutic transfer genes into the genome to maintain stability in the cell may affect gene expression and its unwanted effects on nearby genes. In addition, some genes need to be more significant to transfer by vectors quickly. Finally, foreign genes cannot always be introduced directly into dominant mutations or defective genetic material. To solve the problems related to these basic limitations, conventional methods have emerged to make precise and targeted changes in the genome [12, 13]. In this regard, genome editing is a practical, versatile, and preferred tool for functional gene research, gene therapies, and precise breeding of crops and domestic and attractive animals for practical and industrial research [14, 15].
Genome editing research started in the 1970s. The first major step in gene editing was achieved when researchers showed that when a piece of DNA enters a cell, it can enter the host genome through homologous recombination and implement the desired changes in the cell. This development came when it was found that in eukaryotic cells, more precise gene targeting mechanisms could be achieved by inducing a double-strand break in a specific genomic target. In addition, the scientists found that if a synthetic DNA restriction enzyme was introduced into the cell, it could recognize the DNA at specific locations and cut it into double strands, subsequently repaired by HDR (Homology-Directed Repair) and NHEJ (Non-Homologous End-Joining) mechanisms resulting in homology-based insertions, deletions, or repairs [16, 17]. Among the different methods and mechanisms of gene editing, CRISPR-Cas9 technology (the fourth generation of gene editing) has surpassed other methods. Two female scientists introduced this technology to the scientific community in 2012, making them proud to receive the Nobel Prize in Chemistry in 2020. It is necessary to explain that some sources have announced the beginning of the first research related to gene editing in 1987, which indicates the existence of initial ideas in this regard. However, its expansion has been observed since 2000. CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeat, which has been mentioned in various sources. The CRISPR-Cas9 system was developed after other gene editing systems called meganucleases (a class of endodeoxyribonucleases), ZFNs (Zinc-Finger Nucleases) and TALENs (Transcription Activator-Like Effector Nucleases) due to its unique features, it attracted the attention of researchers, so the publication of articles and inventions in this field shows great growth (Figure 1). It should be noted that most of the research in this regard is dedicated to cancer, AIDS, and hepatitis [18, 19].
MECHANISMS of GENE EDITING
Gene editing can change an organism's DNA sequence, essentially engineering its genetic makeup. This process is carried out using enzymes, specifically nucleases that are engineered to target a specific DNA sequence. They introduce cuts into the DNA strands, allowing the removal of existing DNA and inserting replacement DNA. In other words, the gene editing tools developed today can create double-stranded breaks in the genome, and by repairing these breaks, the process of gene editing can be developed. There are four methods of gene editing: gene destruction or mutation, gene deletion, gene modification and gene insertion. Based on this, researchers use different tools capable of creating double-strand breaks in DNA to create various changes in the genome. Specific nucleases for gene editing include engineered target sequences and restriction enzymes. After the programmed nuclease cleaves the target gene to introduce double-strand breaks (DSBs), molecular repair proceeds via two fundamentally different mechanisms: homology-directed repair (HDR), in which broken DNA is repaired using a homologous DNA sequence as a template, and non-homologous end joining (NHEJ), in which broken ends in a non-homologous DNA sequence are rejoined. The HDR repair mechanism, which allows the insertion of a template DNA to correct or insert a particular sequence at the site of a DNA break, facilitates accurate copying of the template to a specific location in the genome and repairs the homologous DNA break.
In contrast, the NHEJ repair mechanism leads to small insertions or deletions (indels) at the desired site or breaks [20, 21]. As a result, this mechanism can be an efficient way for defective genes to function. As mentioned, today, four different types of nucleases that bind to DNA are used in gene editing: Meganucleases, Zinc-Finger Nucleases (ZFN), Transcription Activator-Like Effector Nucleases (TALEN) and Cas9 Nuclease, which is the most recently discovered type [22, 23]. Table 1 compares these four nucleases regarding features and mechanism of action. Their performance is also shown in Figure 2.
ADVANTAGES of GENE EDITING TECHNOLOGY
Gene editing is essential and valuable in various industrial and research fields. In the continuation of the recent exciting developments in the ease of use, features and characteristics of gene editing technology and their application in different fields are examined [24-26].
Human Health: Gene editing technology creates a fundamental change in gene therapy. It can treat a wide range of diseases (such as diabetes, cancer, cystic fibrosis and sickle cell anaemia) that have not been possible to treat so far using this technology. All cancers result from numerous diverse mutations that lead to the excessive growth and proliferation of cells and the emergence of malignant phenotypes. The site of the event and the area disrupted by these mutations can be classified into four categories: oncogenes, tumor suppressors, epigenetic factors and chemotherapy resistance genes. CRISPR-Cas9 technology, as a powerful tool with high characteristics, can correct these mutations and treat cancers derived from them. Since oncogenic changes in several cancers lead to increased cell proliferation and malignancy, oncogenes such as tyrosine kinase receptor Erb2 can be directly targeted by CRISPR-Cas9 technology. From a complementary point of view, the CRISPR-Cas9 method can create cancer-causing mutations in human cell lines and animal models. In this regard, lung cancer cell lines, acute myeloid leukaemia, and liver and pancreatic cancer have been developed. Crisper-Cas9 technology can also be used in animal models suffering from various diseases (from hereditary diseases to cancers). It has created heritable changes by CRISPR-Cas9 technology and direct targeting of one or more bilateral in the animal egg. Among the transgenic animal models, most of the tests are based on mouse models. However, researchers have succeeded in making models of non-human primates by targeting multiple genes. The advantage of these models is in the reconstruction and the possibility of investigating complex human diseases such as neurodegenerative diseases. However, mouse models have more advantages than others, such as the cost of working with them, and in addition, mouse models are very suitable for extensive mutagenesis studies in vivo [25].
New Materials: By using these technologies, it is possible to achieve the synthesis of new materials that can be used in various applications, such as the release of oral drugs or the production of biosensors.
Drug Development: Engineered cells can be produced using these technologies that produce drug production optimally and efficiently. In addition, it significantly reduces the cost of the drug and provides easy access to the drug.
Research Applications: With technologies such as gene editing, new animals and cell models can be designed and produced, which will help us learn more about diseases and test new drugs and vaccines on those cell and animal models.
Agriculture: By using gene editing tools, it is possible to modify the seeds of agricultural products without harming other genes. Based on this, it is possible to obtain agricultural products that can be resistant to infections and environmental damage, and as a result, food security can be improved.
Bioenergy: With the help of tools such as gene editing, it is possible to produce biofuels (green). Therefore, it is possible to increase and optimize the production of biofuels such as ethanol in algae cells or seeds by modifying the metabolic and biochemical pathways of the relevant cells [24-26].
Criminology: By combining CRISPR technology with DNA fingerprinting technology, new methods can be developed in criminal identity detection and criminology. DNA (genetic) fingerprinting is a method that was first presented to the scientific community and criminology experts in 1985 by Alec Jeffrey using repeatable variable sequences of 15 to 100 games called VNTR (Variable Number of Tandem Repeat). Genetic fingerprinting allows specialists to determine the differences and similarities between people based on specific DNA samples. Based on this, people's communication can be observed and identified at crime scenes. According to each person's VNTR sequence, gRNA (gene editing diagnostic tool) can be designed. Based on this, a fluorescently labelled DNA sample from the victim (in a crime) can be prepared and checked for matching with the VNTR sequence of the victim. CRISPR can also be designed to scan DNA or find a specific VNTR. In the CRISPR DNA scan, if CRISPR fails to target the VNTR, it will not bind to it, meaning no fluorescent dye will appear under UV light. However, if scanning is performed and the target is identified and bound, a fluorescence signal is generated, meaning the VNTR can be present in the DNA [27].
RISKS of GENE EDITING TECHNOLOGY
In today's world, the increasing importance of the knowledge of biology as a basic science is undeniable. As a result of deep studies and many investigations, the boundaries of biology and the findings related to the knowledge of nature have been expanded tremendously. The volume of resulting information and its increasing growth cannot be compared to any era. Today, biotechnology, as a branch of biological applications, has progressed more than at any other time. Due to its applications in health, hygiene and the economy, its importance and value have increased daily. These important advances in biotechnology are mainly due to advances in instrumentation and their application in developing the frontiers of biology. The most remarkable developments of this knowledge and technology have been achieved in ecology, genetics, microbiology, molecular biology, biochemistry, cell culture technologies and process engineering. The emergence of the new sciences of genomics, proteomics, bioinformatics, systems biology, synthetic biology and gene editing has also resulted from these developments. On the other hand, today, biology is exposed to hostile abuse as much as chemistry in World War I and physics in World War II. The enormous force of international trade that underpins this basic science has driven it to innovations that, along with its marketable medical value, may also be used for destructive purposes. If a country exploits science and technology in biological fields, it can reveal one of the most serious problems of humanity that it has not faced so far. If the production of a new generation of bioweapons is pursued with force, especially if used to control and conquer humans, it can cause dangerous technological competition. If the force of biotechnology is not politically contained, it will be able to invent scientific methods that will change the way war is conducted and increase the means of civilian sacrifice. It should be noted that the discussions discussed below do not in any way attempt to magnify the possible risks of biotechnology; because such a possibility exists with any other technology, both those that are widely used in societies today (such as IT and telecommunications) and those that will emerge in the future in the field of health (such as gene editing, artificial synthesis, and human creation). Such studies can also show the potential capabilities of biotechnology in promoting new defense capabilities; so that the guardians of the country's biological defense pay special attention to the potential of this technology for defense purposes (such as providing new treatment trends and new diagnostic methods in the direction of biological defense).
Among the various technologies proposed in the field of life and health today, gene editing technology can cause irreparable risks to the biological security of societies. Now genome editing is much easier, faster, cheaper and more efficient than ever before (just like editing an article on a computer) and helps researchers in various scientific fields. The fourth generation of gene editing, CRISPR-Cas9, can handle new editing programs, from viruses and bacteria to animals, plants and humans. However, as this technology develops, how should it be controlled? While expressing concern about the negative consequences of the development of gene editing technology, Professor Jennifer Doudna, the discoverer of Cas9 nuclease and 2020 Nobel Prize winner, has received $3.3 million in funding from the US Defense Advanced Research Projects Agency (DARPA) to investigate anti-CRISPR solutions. The red line of this technology is the manipulation of the human embryo [28-30].
It should be remembered that CRISPR-based genome editing technology has caused a tremendous revolution in medical sciences and other scientific fields. This technology, developed for about a decade, has fascinated scientists. In a way that it allows a person with less than a high school education to edit the genome of any animal or plant. High school students are now using this technology to perform experiments that were previously only dreams for most scientists. Much of the scientists' research has focused on the tremendous potential of using genome editing to treat cancer. This work is based on the familiarity and understanding of the new model of the cancer network, which examines how cancer cells are controlled. New gene-based approaches for cancer treatment have suggested using genome editing as an effective tool. Unfortunately, while we can cure cancer using CRISPR, we can also create cancer using the same technology.
Based on this, researchers will have been able to design cancer models on the computer. The possibility of artificially creating cancerous tumors will also be provided when they can treat cancer patients with CRISPR. Therefore, the cancer bomb can be implemented using CRISPR. As a result, today, CRISPR technology is proposed as a new biological weapon, and scientists warn about its biosecurity consequences [31, 32]. The very simple application of CRISPR makes this technology potentially very dangerous. This technology has features that make it an ideal military and bioterrorist weapon and has been favored by biohackers. Mr. Josiah Zayner, one of the pioneers of biohacking and the founder of Odin Company since 2006, offers online training and simple gene therapy kits based on CRISPR technology to his customers around the world, who, in addition to the simple training of gene editing, can design and produce genetically engineered products (such as the green tree frog with the high growth rate) [33].
Based on this, it seems that in the not-too-distant future, we will see the design and production of biological weapons (mass destruction) based on CRISPR, which will be even more dangerous than a nuclear bomb. According to some scientists, nuclear weapons are obsolete because they are complicated to maintain. From a military point of view, CRISPR weapons are considered far superior to nuclear weapons and will probably replace them. These CRISPR-based editing features make it very easy to weaponize. It is possible to create an engineered virus that edits CRISPR in a completely controlled manner so that only humans whose genomes have specific characteristics are killed or disabled by the virus. The importance of CRISPR biosecurity is so high that scientists have warned about the design and production of programmed humans and editing on human embryos and are drafting national and international laws in this regard. It is necessary to explain that the Chinese scientist, He Jiankui, on October 8, 2018, officially announced the birth of Chinese girl twins with Crisper technology. In these twins, who were born from a father with AIDS and a healthy mother, the CCR5 gene, which is related to the entry of HIV into cells, has been edited and deleted. This scientific activity, carried out illegally and confidentially, shocked the scientists in this field and led to a fine and three years of imprisonment for this Chinese scientist. Jiankui had to present the result of his research at the Second International Conference on Human Genome Editing held in Hong Kong in 2018, which was widely criticized by the scientific community, even Chinese scientists [34].
The question raised today is why the Crisper weapon can be so dangerous. The answer to this question can be found in the following:
1- CRISPR-Cas based editing can be precisely designed to edit a specific part of the target genome.
2- Viruses can deliver CRISPR-Cas-based editing to a given host.
3- Mathematical rules can fully control edits. In other words, target genome editing is applied only if certain precise conditions are present in the target person's genome. For example, two people can be infected by a CRISPR-based editing virus, but only the person with the prerequisites will have their genome edited. In addition to these, unfortunately, many more characteristics of a potential CRISPR-based weapon make them ideal weapons for future precision and targeted mass destruction. For example, its effects can remain hidden for months, and CRISPR bombs do not have the long-term toxic effects of nuclear weapons. Also, it can be implemented for most organisms and programmed as an effective weapon for the nervous system. As a result, CRISPR as a weapon of mass destruction for genocide is significant and considered [34, 35]. Also, from a biosecurity perspective, CRISPR technology could potentially create and induce precise cancers that would kill people within months. Unfortunately, it is much easier to cause cancer than to cure it with genome editing. The main appeal of a deadly CRISPR-engineered virus as a bioterrorism weapon is the precision in mass incapacitation and destruction of people. Unfortunately, these topics are unrelated to science fiction and fantasy horror movies, but this is a real danger in humanity's present and future. Notably, there are probably labs worldwide developing CRISPR technology to develop the next generation of bioweapons. As mentioned, Crisper also has genocidal capabilities. Given that a particular generation has unique genetic traits that distinguish it from others, all members are potential targets for a CRISPR-engineered killer virus. For example, if the prerequisite is that one must have brown eyes, then anyone with brown eyes is a potential target for a deadly CRISPR-engineered virus. Using the Crisper weapon, you can create diseases that cause the target person to die slowly or quickly. Some scientists who have provided significant analysis about the possible possibilities have raised various dangerous issues. Some of these reports indicate major future concerns that could result from secret research programs. Especially these reports have not ruled out the possibility of hidden viruses that can secretly enter the genome of a population and later be activated by a signal. Another example is "programmed cell death". This ability to insert a genome into the genetic reserve of a specific population and attack it at will, or to create a completely new pathogenic agent, indicates a change in capabilities [35, 36].
Gene Editing Defence Strategies
On June 29, 2018, in Newsweek magazine, the media announced and published the discussion of using gene editing technology on human embryos to eliminate genetic diseases. However, some scientists and pioneers of this technology (such as Professor Doudna) raise ethical considerations as a serious discussion. Since 2015, China has started extensive research on human embryos under the leadership of Professor Lu You, an oncologist at Sichuan University in Chengdu, and is still developing this technology on human embryos; in a way that on October 28, 2017, a group led by him injected the modified cells into a patient with aggressive lung cancer as part of a clinical trial at the West China Hospital [37]. However, according to most scientists, the red line of gene editing technology is the genetic manipulation of human embryos. However, today, some scientists secretly use gene editing technology to research and develop human embryos to design and create disease-free humans with special capabilities. These designed children have high intelligence, creative mind, five senses with great power and are resistant to various diseases. Also, some have called this technology the engine of creation because it gives the scientist a god-like power (based on his beliefs) to create and improve future humans (super-humanity). On the other hand, unlike the Schilling Act, CRISPR-based bioweapons have international strategic implications. According to Thomas Schelling's Game Theory, a balanced international strategy is observed in the nuclear age. If one side increases or decreases its nuclear weapons capabilities to maintain balance, the other side must follow suit. Based on this, each side knows what weapons the other side has, and both sides, knowing this (based on open inspection of nuclear facilities), observe a balance in this regard. However, in the era of genome editing technology, due to the ease of making CRISPR-based biological weapons in small laboratories that are impossible to observe and identify, the awareness of the ability and capacity to design and manufacture biological weapons on the other side has failed and as a result, the strategy based on inspection and mutual destruction of such weapons of mass destruction also fails. Therefore, CRISPR-based bioweapons represent a fundamental change in Schilling's Law. A balanced, no-win, bargaining, nonzero-sum Schilling game approaches the classical zero-sum game, in a way that the side that attacks first may win the entire game. As a result, according to Schelling's argument, this international situation will be very unstable, fragile and dangerous [35].
Based on this, how can the defense strategy to deal with the threats resulting from such technologies and biological security be? In other words, how do we defend ourselves against biological weapons caused by CRISPR? To answer these questions, it is important to pay attention to the following:
1- The attention and awareness of public opinion and political leaders should be increased about the dangers of CRISPR-based genome editing.
2- Considering the actual and potential risks of CRISPR-based genome editing in bioterrorist attacks, defense strategies should be quickly developed to deal with such possible attacks.
3- Any CRISPR-based editing can be reversed. In other words, a network mutation that causes cancer can be reversed to stop cancer. When we know how to stop cancer, we will know how to cause cancer. Alternatively, any gene essential for life can be disabled by a reverse edit. This requires a coherent defense-research initiative.
4- It is necessary to adopt coherent international laws (in line with the Convention on the Prohibition of the Development, Production and Stockpiling of Biological Weapons) to examine the risks of this very dangerous technology (in order to confront and not prevent it).
5- A new international bioethics committee is needed to deal with risk groups in this regard.
Conclusion
Gene editing technology has shown the scientific community a new window and solution through which appropriate treatment methods can be adopted to deal with most diseases. Therefore, genome editing offers great opportunities in biology, biotechnology and medical sciences, including preventing and treating diseases and producing good food. On the other hand, CRISPR-based bioweapons destroy the logical and strategic balance of power that has kept the world free of catastrophic wars. The world is facing a potentially more dangerous technology than nuclear weapons because of its ease of development and precision of use. Precise targeting of individuals with a deadly CRISPR-based virus means that, as in nuclear war, there is no longer a barrier to mutually assured destruction. Instead, we are faced with the possibility of a precise and targeted mass genocide. On the other hand, after the birth of the Chinese gene-edited twins, in terms of Islamic jurisprudence and ethics, there have been discussions about the possibility of using CRISPR technology to develop embryonic research and human genome editing. Considering that embryonic gene editing may lead to hereditary changes in the human genome, whether this practice should be permissible requires a deep and detailed discussion from different perspectives. Islam's views on the concerns raised about human genome editing consider the moral principles important in Islam and declare that it should be taken into account when evaluating the permissibility of gene editing of the human reproductive line through CRISPR. As discussed in this article, human embryo editing research for medical purposes is legal under certain conditions and is used to treat diseases, but until the safety and effectiveness issues of this technology are resolved, it should not be applied to humans. Strong and strict ethical guidelines are necessary to preserve human dignity and prevent the misuse of technology, and religious principles of preserving human life, descent and dignity and preventing possible harm are among the important principles in evaluating the permissibility of human embryo editing through CRISPR from an Islamic point of view. Therefore, it can be concluded that human gene editing by CRISPR is considered halal in Islam if it has the following conditions:
A- To be used only for medical purposes, especially for preventing or treating diseases. This kind of change is not considered a manipulation of God's creation.
B- It is allowed only after solving the safety and efficiency problems, and the technology used should not cause more harm to the parents, the resulting child, the society and the future generation.
C- Establish strict regulations to ensure respect for the people involved, prevent early use and abuse of technology, and seriously prevent unwanted genetic changes in humans [38-40].
Clinical & Practical Tips in POLICE MEDICINE: Considering the emergence of promising technologies such as genome editing, in addition to using the opportunities obtained from this technology, especially in the field of life and health and diagnosis and treatment of incurable diseases, it is necessary to pay special attention to this technology from the point of view of biosecurity. The creation of new organisms, biohacking and the creation of programmed humans are among the future biosecurity risks of this technology, and the Deputy Health and Medical Services of General Staff of the Armed Forces must draw specific plans and strategies in this regard in cooperation with academic centers.
Acknowledgements: This study was carried out in the Research Institute of Biological Sciences and Technology of the Malek Ashtar University of Technology, and the efforts of the officials of this university are gratefully acknowledged.
Conflict of interest: The article's authors stated that the present study has no conflict of interest.
Authors' Contribution: Samane Fethollahi (data collection); Dr Mehdi Zein al-Dini (presenting the idea and design of the study, data collection, data analysis). All the authors participated in the initial writing of the article and its revision, and all accept the responsibility for the accuracy and correctness of the article's contents with the final approval of this article.
Financial Sources: The present study was carried out with the financial support of the Biological Sciences Department of the Academic Complex of Chemistry and Chemical Engineering of Malek Ashtar University of Technology, in line with the observation of emerging technologies, and was a part of the research project number "19730180208".
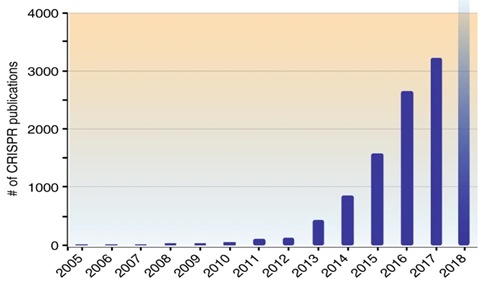
Figure 1) The amount of published articles related to gene editing from 2005 to 2018
Table 1) Comparison of different genome editing methods
| Nuclease Mechanism of action |
Meganuclease | ZFNs | TALEN | CRISPR/Cas9 |
| DNA system | Protein-DNA | Protein-DNA | Protein-DNA | RNA-DNA |
| Synonymous with targeting | 12-45 games | 18-36 games | 30-40 games | 22 games |
| Price | Much | Much | medium | Low |
| Off-target events | Low | Comparable | Comparable | Comparable |
| release therapy | simple | simple | complicated | medium |
| Multiple targeting | complicated | complicated | complicated | simple |
| specificity (off target) | Very specific | Relatively non-specific | exclusive | Relatively non-specific |
| Requires dimerization | no | Yes | Yes | no |
| Vector packaging | simple | complicated | complicated | medium |
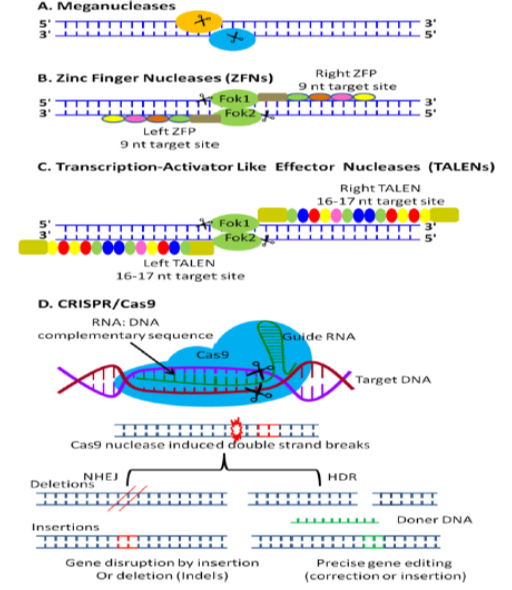
Figure 2) A picture of the mechanism of targeted nucleases for gene editing. (A) meganuclease, (B) ZFNs, (C) TALENs, (D) gene editing process using CRISPR/Cas9 technology
Article Type: Analytic Review |
Subject:
Police Medicine Related Technologies
Received: 2023/01/15 | Accepted: 2023/02/20 | Published: 2023/04/8
Received: 2023/01/15 | Accepted: 2023/02/20 | Published: 2023/04/8
References
1. Riedel S. Biological warfare and bioterrorism: A historical review. J Baylor Scott White Health. 2004;17(4):400-06. doi:10.1080/08998280.2004.11928002 [DOI:10.1080/08998280.2004.11928002] [PMID] [PMCID]
2. Barras V., Greub G. History of biological warfare and bioterrorism. Clin Microbiol Infect. 2014;20(6):497-502. doi: 10.1111/1469-0691.12706 [DOI:10.1111/1469-0691.12706] [PMID]
3. Xue Y, Yu H, Qin G. Towards good governance on dual use biotechnology for global sustainable development. Sustainability. 2021;13:14056. doi: 10.3390/su132414056 [DOI:10.3390/su132414056]
4. DaSilva EJ. Biological warfare, bioterrorism, biodefense and the biological and toxin weapons convention. Elect J Biotechnol. 1999;2(3). doi: 10.2225/vol2-issue3-fulltext-2 [DOI:10.2225/vol2-issue3-fulltext-2]
5. DiEuliis D. Key national security questions for the future of synthetic biology. Fletcher Forum World Aff. 2019;43:127-40. https://www.jstor.org/stable/45289832
6. National Research Council. Biotechnology Research in an Age of Terrorism; National Academies Press: Washington, DC, USA, pp. 16-17, 2004. doi:10.17226/10827 [DOI:10.17226/10827]
7. Casadevall A. The future of biological warfare. Microb Biotechnol. 2012;5(5):584-87. doi: 10.1111/j.1751-7915.2012.00340.x. [DOI:10.1111/j.1751-7915.2012.00340.x] [PMID] [PMCID]
8. Aken JV., Hammond E. Genetic engineering and biological weapons. EMBO Reports. 2003;4:S57-S60. [DOI:10.1038%2Fsj.embor.embor860]
9. Black JL 3rd. Genome projects and gene therapy: gateways to next generation biological weapons. Mill Med. 2003;168(11):864-71. https://pubmed.ncbi.nlm.nih.gov/14680038/ [DOI:10.1093/milmed/168.11.864] [PMID]
10. Fraser CM. A genomics-based approach to biodefence preparedness. Nat Rev Gen. 2004;5:23-33. doi: 10.1038/nrg1245 [DOI:10.1038/nrg1245] [PMID] [PMCID]
11. Carrasco-Ramiro F, Peiro-Pastor R, Aguado B. Human genomics projects and precision medicine. Gene Ther. 2017;24:551-61. doi:10.1038/gt.2017.77 [DOI:10.1038/gt.2017.77] [PMID]
12. Goswami R, Subramanian G, Silayeva L, Newkirk I, Doctor D, Chawla K et al. Gene therapy leaves a vicious cycle. Front Oncol. 2019;9:297. doi: 10.3389/fonc.2019.00297 [DOI:10.3389/fonc.2019.00297] [PMID] [PMCID]
13. Pfeifer A, Verma IM. Gene therapy: promises and problems. Annu Rev Genomics Hum Genet. 2001;2:177-211. doi: 10.1146/annurev.genom.2.1.177. PMID: 11701648. [DOI:10.1146/annurev.genom.2.1.177] [PMID]
14. Ahmar S, Saeed S, Khan MHU, Ullah Khan S, Mora-Poblete F, Kamran M et al. A revolution toward gene-editing technology and its application to crop improvement. Int J Mol Sci. 2020;21(16):5665. doi: 10.3390/ijms21165665. [DOI:10.3390/ijms21165665] [PMID] [PMCID]
15. Li C, Brant E, Budak H, Zhang B. CRISPR/Cas: a nobel prize award-winning precise genome editing technology for gene therapy and crop improvement. J Zhejiang Univ Sci B.2021;22(4):253-84. doi:10.1631/jzus.B2100009 [DOI:10.1631/jzus.B2100009] [PMID] [PMCID]
16. Khalil A.M. The genome editing revolution: review. J Genet Eng Biotechnol. 2020;18(1):68. doi:10.1186/s43141-020-00078-y [DOI:10.1186/s43141-020-00078-y] [PMID] [PMCID]
17. Miyaoka Y, Berman JR, Cooper SB, Mayerl SJ, Chan AH, Zhang B et al. Systematic quantification of HDR and NHEJ reveals effects of locus, nuclease and cell type on genome editing. Sci Rep. 2016;6:23549. doi:10.1038/srep23549 [DOI:10.1038/srep23549] [PMID] [PMCID]
18. Adli M. The CRISPR tool kit for genome editing and beyond. Nat Commun. 2018;9(1):1911. doi: 10.1038/s41467-018-04252-2. [DOI:10.1038/s41467-018-04252-2] [PMID] [PMCID]
19. Li H, Yang Y, Hong W, Huang M, Wu M, Zhao X. Applications of genome editing technology in the targeted therapy of human diseases: mechanisms, advances and prospects. Sig Trans Targ Ther. 2020;5:1.doi:10.1038/s41392-019-0089-y [DOI:10.1038/s41392-019-0089-y] [PMID] [PMCID]
20. Maeder M, Gersbach C. Genome-editing technologies for gene and cell therapy. Molecular Therapy. 2016;24(3):430-46. doi: 10.1038/mt.2016.10. [DOI:10.1038/mt.2016.10] [PMID] [PMCID]
21. Kim H; Kim J. A guide to genome engineering with programmable nucleases. Nature Reviews Genetics. 2014;15(5):321-34. doi: 10.1038/nrg3686. [DOI:10.1038/nrg3686] [PMID]
22. Abbas Raza SH, Hassanin AA, Pant SD, Bing S, Sitohy MZ, Abdelnour SA. et al. Potentials, prospects and applications of genome editing technologies in livestock production. Saudi J Biol Sci. 2022;29:1928-35. DOI: 10.1016/j.sjbs.2021.11.037 [DOI:10.1016/j.sjbs.2021.11.037] [PMID] [PMCID]
23. Guha TK, Edgell DR. Application of alternative nucleases in the age of CRISPR/Cas9. Int J Mol Sci.2017;18:2565. doi:10.3390/ijms18122565. [DOI:10.3390/ijms18122565] [PMID] [PMCID]
24. Barrangou R., Sontheimer EJ., Marraffini LA. CRISPR: Biology and application. John Wiley & Sons Inc. 2022. 304p. https://www.wiley.com/en-au/CRISPR%3A+Biology+and+Applications-p-9781683673613 [DOI:10.1002/9781683673798]
25. Addgene, CRISPR 101, Synthego, 2017. www.addgene.org.
26. Kaboli S and Babazada H. CRISPR mediated genome engineering and its application in industry. Curr Issues Mol Biol, 2018;26:81-92. doi: 10.21775/cimb.026.081 [DOI:10.21775/cimb.026.081] [PMID]
27. Masood U. DNA Fingerprinting and CRISPR cas9 System. Eur Exp Biol. 2021;11(5):138. https://www.primescholars.com/articles/dna-fingerprinting-and-crispr-cas9-system.pdf
28. Isaacson W. The code breaker: Jennifer Doudna, Gene editing, and the future of the human race. Simon & Schuster Books for Young Readers. 2022. 560p. https://www.amazon.com/Code-Breaker-Jennifer-Doudna-Editing/dp/1982115858
29. Carey N. Hacking the code of life: How gene editing will rewrite our futures. Icon Books; 2019. 176p. https://www.amazon.com/Hacking-Code-Life-editing-rewrite/dp/1785784978
30. Doudna JA, Sternberg SH. A crack in creation: Gene editing and the unthinkable power to control evolution. Mariner Books. 2017. 304p. https://www.amazon.com/Crack-Creation-Editing-Unthinkable-Evolution/dp/0544716949
31. West RM, Gronvall GK. CRISPR cautions: Biosecurity implications of gene editing. Perspect Biol Med. 2020;63(1):73-92. doi: 10.1353/pbm.2020.0006. PMID: 32063588. [DOI:10.1353/pbm.2020.0006] [PMID]
32. Vogel KM, Ouagrham-Gormley SB. Anticipating emerging biotechnology threats: A case study of CRISPR. Politics Life Sci. 2018;37(2):203-219. doi: 10.1017/pls.2018.21. PMID: 31120699. [DOI:10.1017/pls.2018.21] [PMID]
33. DiEuliis D and J. Giordano, Gene editing using CRISPR/Cas9: implications for dual-use and biosecurity. Protein Cell. 2018;9(3):239-40. doi:10.1007/s13238-017-0493-4 [DOI:10.1007/s13238-017-0493-4] [PMID] [PMCID]
34. Alonso M, Savulescu J. He Jiankuis gene-editing experiment and the non-identity problem. Bioethics. 2021;00:1-11. DOI:10.1111/bioe.12878 [DOI:10.1111/bioe.12878] [PMID] [PMCID]
35. Werner E. The coming CRISPR wars: Or why genome editing can be more dangerous than nuclear weapons. Preprint. 2019. doi: 10.13140/RG.2.2.17533.00485
36. DiEuliis D and Giordano J. Why gene editors like CRISPR/Cas may be a game-changer for Neuroweapons. Health Secur. 2017;15(3):296-302. DOI:10.1089/hs.2016.0120 [DOI:10.1089/hs.2016.0120] [PMID] [PMCID]
37. Cyranoski D. CRISPR gene-editing tested in a person for the first time. Nature. 2016;539:479. [DOI:10.1038/nature.2016.20988] [PMID]
38. Munirah Isa N, Zulkifli NA, Man S. Islamic perspectives on CRISPR/Cas9-mediated human germline gene editing: A preliminary discussion. Sci Eng Ethics. 2020;26:309-23. doi:10.1007/s11948-019-00098-z [DOI:10.1007/s11948-019-00098-z] [PMID]
39. Alsomali N, Hussein G. CRISPR-Cas9 and He Jiankuis case: an Islamic bioethics review using Magasid al-Sharia and Qawaid Fighiyyah. Asian Bioethics Rev. 2021;13:149-65. doi.org/10.1007/s41649-021-00167-1 [DOI:10.1007/s41649-021-00167-1] [PMID] [PMCID]
40. Al-Balas QAE. Dajani R., Al-Delaimy WK. The ethics of gene editing from an islamic perspective: A focus on the recent gene editing of the chinese twins. Sci Eng Ethics. 2020;1851-60. doi.org/10.1007/s11948-020-00205-5 [DOI:10.1007/s11948-020-00205-5] [PMID]
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |

