
نشریه علمی پژوهشی طب انتظامی Journal of Police Medicine

Volume 11, Issue 1 (2022)
J Police Med 2022, 11(1) |
Back to browse issues page
Ethics code: (IR.IAU.ARAK.REC).1399.009
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Soleimani H, Malekirad A, Mohajerani H R, jafari P, Akbari N. The Effect of Probiotic Dietary Supplementation on Ethanol-Induced White and Grey Matter Damage to the Brain in Male Wistar Rats. J Police Med 2022; 11 (1)
URL: http://jpmed.ir/article-1-1076-en.html
URL: http://jpmed.ir/article-1-1076-en.html
1- Department of Biology, Faculty of Basic Sciences, Arak Branch, Islamic Azad University, Arak, Iran
2- Department of Biology & Psychology, Faculty of Basic Sciences, Payame Noor University, Tehran, Iran
3- Applied Neuroscience Research Center, Islamic Azad University, Arak, Iran ,mohajeranihr@gmail.com
4- Department of Biology, Faculty of Basic Sciences, Arak Branch, Islamic Azad University, Arak, Iran, Arak
2- Department of Biology & Psychology, Faculty of Basic Sciences, Payame Noor University, Tehran, Iran
3- Applied Neuroscience Research Center, Islamic Azad University, Arak, Iran ,
4- Department of Biology, Faculty of Basic Sciences, Arak Branch, Islamic Azad University, Arak, Iran, Arak
English Extended Abstract: (2842 Views)
INTRODUCTION
Ethanol is a very strong pro-oxidant that causes cellular oxidative damage, and most of its dangerous effects are attributed to acetaldehyde [1]. ... [2, 3]. Alcohol addiction is usually one of the most challenging cases that traffic police and police headquarter deal it. Many criminal behaviors occur while intoxicated [4]. ... [5]. Evidence shows that ethanol exerts its effects on the nervous system by direct or indirect effects on various neurotransmitter systems, especially acetylcholine, dopamine, Gabapentin, glutamate, and amino acid neurotransmitters [6]. ... [7-9]. Probiotics are live microorganisms that prevent several harmful effects of neurodegeneration such as neurotransmitter depletion, chronic inflammation, oxidative stress, and programmed cell death [10]. Research findings show that the effect of probiotics on improving memory in behavioral tests and its prescription does not affect synaptic activity [11]. Also, the significance of the predictive power of probiotic consumption to reduce depression does not apply to anxiety [12].
AIM(s)
The present study was designed to investigate the variant effect of probiotics as a food supplement, which is effective in improving neural activities in previous studies [10], on white and gray matter damage caused by ethanol in the brain of male rats.
RESEARCH TYPE
This is an experimental and case-control study.
RESEARCH SOCIETY & PLACE
Male Wistar rats were used in this study, which was conducted in the Physiology Research Laboratory of Arak, Iran Islamic Azad University.
SAMPLING METHOD & NUMBER
The sample size was based on the formula for determining the number of samples in case-control experimental studies, and 32 adult mice (12 weeks ) with an average weight of 250-300 grams were selected. The samples were randomly divided into four groups of eight. 1) Negative control group in which mice were gavaged with only one milliliter of phosphate buffer for two months. 2) Probiotic group in which Lactobacillus casei with a concentration of 10⁹cfu/ml was gavage for two months. 3) In The group receiving ethanol that one milliliter of phosphate buffer was gavage for two months and ethanol (2g/kg) was injected intraperitoneally every day during the last five days of the period. 4) Probiotic-ethanol group, which was initially gavage with Lactobacillus casei with a concentration of 10⁹cfu/ml for two months, and ethanol injection was performed simultaneously on the last five days of the probiotic gavage period.
USED DEVICES & MATERIALS
The tool used in this research was a histological and tissue staining set. The probiotics used in this study were prepared by a biotechnology company (Takgene- group) and were guaranteed to contain at least 1 CFU/ml ×10⁹ Lactobacillus casei.
METHODS
All groups were intervened at the same time (at eight o'clock in the morning every day) on all days for two months so that groups (I) and (III) received phosphate buffer and groups (II) and (IV) received probiotics. In addition, in groups (III) and (IV), ethanol was injected every day on the same day in the last five days of this period. In all groups, at 20:00 on the last day of the intervention, the animals were anesthetized and sacrificed, and subjected to histopathological study. To perform histological studies in the target areas, cardiac perfusion was performed after half of the animals in each group were anesthetized with a mixture of ketamine hydrochloride (50mg/kg) and xylazine (5mg/kg). After the completion of perfusion, the brain was carefully removed from the skull, and after removing the brain, it was placed in containers containing 10% formalin to fix the sample. Then the sample was cut and the prepared cut was placed inside the cassette or basket and the lid was closed and placed inside the special basket of the PROCESSOR device. The time was set to be 18 hours in the machine to fix the sample, dehydrate and clarify. This device included 12 containers, two formalin, six alcohol 50-100, and two xylol containers, and finally, two containers containing 60-degree paraffin (because paraffin hardens the sample at a temperature above 65 degrees and a temperature lower than 60 not melt grade). After the PROCESSOR finished working, the samples were removed from the machine and the blocking process started. At first, the containers intended for pouring paraffin were placed on the HOT PLATE device to heat the containers and bring them to the same temperature as the sample. Then, using a DISPENSER device, paraffin was poured into metal containers. The lid of the cassettes was opened and with the help of tweezers we put the sample in the container containing paraffin and the lid was closed. In this way, the blocks were prepared and ready to be cut. Sectioning of brain tissues containing black and white brain matter around the lateral ventricles was done according to the Paxinos and Watson atlas. The samples were cut using a microtome. After preparing the main cut, we put it in a hot water bath and removed it from the surface of the water using a slide. Then, the slide containing the section of the desired sample was placed in a special basket for staining. In the next step, we first put the slides in a 100-degree oven for 15 to 20 minutes to melt the excess paraffin and remove it from the sample. In the next step, we placed the samples in Xylenol to completely remove the excess paraffin from the samples (deparaffinization step). Then the samples were dried and placed in 70% alcohol for six minutes. In the next step, washing with distilled water was done, then they were placed in hematoxylin dye for 6 minutes. Then washing was done and in the next step, it was placed in eosin dye for 6 minutes. Then washing was done and then it was placed in 70% alcohol and then 90% alcohol thus the slides were prepared and microscopic images were taken with an optical microscope (Japan Olympus-BH2) and pathological markers were observed under the optical microscope.
ETHICAL PERMISSIONS
The method of this research was approved by the Medical Ethics Committee of Arak, Iran Azad University with code IR.IAU.ARAK.REC.1399.009.
STATISTICAL ANALYSIS
The analysis of the results was done based on the qualitative changes in the brain matters and the comparison of the sections of the experimental groups in terms of cell ischemia, vasogenic and hydrostatic parameters, and the typical microscopic slides were qualitatively compared. To compare the matter sections in terms of the amount and type of lesions, the scoring method was used by including zero (no damage) to four (maximum damage) and calculating the average damage in each group. Prism-GraphPad 9 software was used to analyze data and draw graphs. The comparison of groups was done with a one-way analysis of variance. Tukey's post-test was also used to determine the difference between each group. p value<0.05 was considered as the criterion of the significance level of difference between groups.
FINDING by TEXT
Microscopic examinations in the group receiving pure ethanol showed numerous pathological changes. Vasogenic edema, which could be seen with the strong expansion of the Virchow-Robin spaces as shown in Figure 1, this pathological change was caused by the effects of ethanol in the blood/brain barrier, and by increasing the intracranial pressure and reducing the blood supply to the neuronal cells, which are very sensitive to lack of oxygen and it leads to Cell damage changes. The difference between experimental groups in terms of vasogenic edema was significant (Figure 1); So the ethanol group showed the most damage compared to the negative control group (p<0.0001), while probiotic consumption reduced this damage compared to the ethanol group (p<0.01). Hydrostatic edema, which occurred due to the inflammation of nerve tissue and the blockage of the flow of cerebrospinal fluid in the cerebral ventricles, was visible as edema in the periphery of the cerebral ventricles (Figure 1). Also, the statistical comparison showed a significant difference between the ethanol group and the negative control group in terms of this injury (p<0.0001). Probiotic was able to reduce the significant difference in the damage caused by ethanol with the negative control group (p<0.0001). The results of this statistical comparison are shown in chart 2. Central chromatolysis in brain neurons was visible in the form of cell swelling, moving the nucleus to the cell periphery, and scattering of Nissl bodies (Figure 1). Neuronal ischemic damage was caused by blood supply disruption and caused a significant difference in this respect between the ethanol group and the negative control group (p<0.0001), although the use of probiotics along with ethanol could not compensate for this damage compared to the negative control group. But it caused a significant decrease in this regard compared to the ethanol group (p<0.01) (Chart 3). In these pathological changes, the neuronal cells were shriveled in a triangular shape with pyknosis nuclei that lost their central polarity in the cell cytoplasm (Figures 1 and 2). Following the spread of nerve tissue damage, the accumulation of glial cells was another pathological finding that indicated the extreme damage of nerve tissue under the influence of pure ethanol (Figure 3). In the group receiving ethanol along with probiotics, the changes were reduced by 15-25% compared to the group receiving pure ethanol, and the healthy groups and only probiotics had a healthy tissue pattern (Figure 4).
MAIN COMPARISON to the SIMILAR STUDIES
The healing effects of the probiotic on brain tissue are likely due to its ability to protect against ethanol-induced cellular damage and oxidative stress [13]. The difference between the ethanol and probiotic-ethanol groups may be due to the changes that ethanol has made in the brain tissue, especially around the lateral ventricles. Ethanol has led to cell damage by increasing intracranial pressure and reducing blood flow to hypoxia-sensitive nerve cells. Ischemic neuron damage is caused by blood flow disorder. The formation of significant amounts of brain acetaldehyde in vivo after ethanol consumption and its mechanism is not clearly defined, although catalase is a promising candidate [14]. Dendritic and synaptic changes have been documented in alcoholics, which receptor and transmitter changes, may explain the functional changes and cognitive deficits that result from more strong structural changes [15]. Based on the review of valid databases, this is probably the first study that investigated the effect of probiotics on the above tissue parameters in the acute ethanol administration model and based on the authors' review, there was no similar research to compare the results. But it is possible to analyze the effect of probiotics on the side effects of ethanol that indirectly lead to tissue changes in the brain. ... [16]. Probiotics reduce oxidative stress in alcohol-fed rodents. There is a possibility that this may be due in part to reduced CYP2E1 expression, another study showed that this finding may be the result of the healing of intestinal dysbiosis caused by alcohol (and oxidative stress), as it has been shown the case for probiotics and LactobacillusGG [17]. ... [18-24].
SUGGESTIONS
It is suggested to investigate the effect of nutritional supplements of other valuable probiotic strains on brain damage caused by ethanol. Also, the effect of the probiotic and prebiotic combination on brain poisoning caused by ethanol and other models of brain poisoning are studied.
LIMITATIONS
The limitations of the current research were mainly related to the unavailability of the studied probiotics. Also, due to the time limitation in the use of laboratory facilities, only the model of acute poisoning caused by ethanol was investigated.
CONCLUSIONS
Acute ethanol injection can cause visible pathological changes in the brain tissue, including vasogenic edema with strong expansion of the Virchow-Robin space, hydrostatic edema caused by (swelling or inflammation?) of nerve tissue, and (obstruction or blockage?) of cerebrospinal fluid flow in cerebral ventricles, central chromatolysis and ischemic neuronal damage. It follows disruption in blood supply and the accumulation of glial cells. A probiotic supplement can compensate for the tissue damage caused by ethanol in cortical and subcortical areas adjacent to cerebral ventricles. It appears that probiotics can exert their preventive pathways against alcohol toxicity through biochemical and microflora changes that improve brain tissue.
CLINICAL & PRACTICAL TIPS in POLICE MEDICINE
The use of ethanol causes damage to the white and gray matter of the brain and it is necessary to apply controls against its abuse. The consumption of probiotic food supplements in people under observation or control who suffered from ethanol abuse for any reason can have a compensatory and reducing effect on the damage of white and gray matter in the brain caused by it.
ACKNOWLEDGMENT
The researchers of this study thank TAKGENE Bio Company for providing probiotics in this research.
CONFLICT of INTEREST
The authors of the article state that there is no conflict of interest.
FUNDING SOURCES
This study was supported as a doctoral thesis by Arak, Iran Islamic Azad University.
Tables & Figures
Ethanol is a very strong pro-oxidant that causes cellular oxidative damage, and most of its dangerous effects are attributed to acetaldehyde [1]. ... [2, 3]. Alcohol addiction is usually one of the most challenging cases that traffic police and police headquarter deal it. Many criminal behaviors occur while intoxicated [4]. ... [5]. Evidence shows that ethanol exerts its effects on the nervous system by direct or indirect effects on various neurotransmitter systems, especially acetylcholine, dopamine, Gabapentin, glutamate, and amino acid neurotransmitters [6]. ... [7-9]. Probiotics are live microorganisms that prevent several harmful effects of neurodegeneration such as neurotransmitter depletion, chronic inflammation, oxidative stress, and programmed cell death [10]. Research findings show that the effect of probiotics on improving memory in behavioral tests and its prescription does not affect synaptic activity [11]. Also, the significance of the predictive power of probiotic consumption to reduce depression does not apply to anxiety [12].
AIM(s)
The present study was designed to investigate the variant effect of probiotics as a food supplement, which is effective in improving neural activities in previous studies [10], on white and gray matter damage caused by ethanol in the brain of male rats.
RESEARCH TYPE
This is an experimental and case-control study.
RESEARCH SOCIETY & PLACE
Male Wistar rats were used in this study, which was conducted in the Physiology Research Laboratory of Arak, Iran Islamic Azad University.
SAMPLING METHOD & NUMBER
The sample size was based on the formula for determining the number of samples in case-control experimental studies, and 32 adult mice (12 weeks ) with an average weight of 250-300 grams were selected. The samples were randomly divided into four groups of eight. 1) Negative control group in which mice were gavaged with only one milliliter of phosphate buffer for two months. 2) Probiotic group in which Lactobacillus casei with a concentration of 10⁹cfu/ml was gavage for two months. 3) In The group receiving ethanol that one milliliter of phosphate buffer was gavage for two months and ethanol (2g/kg) was injected intraperitoneally every day during the last five days of the period. 4) Probiotic-ethanol group, which was initially gavage with Lactobacillus casei with a concentration of 10⁹cfu/ml for two months, and ethanol injection was performed simultaneously on the last five days of the probiotic gavage period.
USED DEVICES & MATERIALS
The tool used in this research was a histological and tissue staining set. The probiotics used in this study were prepared by a biotechnology company (Takgene- group) and were guaranteed to contain at least 1 CFU/ml ×10⁹ Lactobacillus casei.
METHODS
All groups were intervened at the same time (at eight o'clock in the morning every day) on all days for two months so that groups (I) and (III) received phosphate buffer and groups (II) and (IV) received probiotics. In addition, in groups (III) and (IV), ethanol was injected every day on the same day in the last five days of this period. In all groups, at 20:00 on the last day of the intervention, the animals were anesthetized and sacrificed, and subjected to histopathological study. To perform histological studies in the target areas, cardiac perfusion was performed after half of the animals in each group were anesthetized with a mixture of ketamine hydrochloride (50mg/kg) and xylazine (5mg/kg). After the completion of perfusion, the brain was carefully removed from the skull, and after removing the brain, it was placed in containers containing 10% formalin to fix the sample. Then the sample was cut and the prepared cut was placed inside the cassette or basket and the lid was closed and placed inside the special basket of the PROCESSOR device. The time was set to be 18 hours in the machine to fix the sample, dehydrate and clarify. This device included 12 containers, two formalin, six alcohol 50-100, and two xylol containers, and finally, two containers containing 60-degree paraffin (because paraffin hardens the sample at a temperature above 65 degrees and a temperature lower than 60 not melt grade). After the PROCESSOR finished working, the samples were removed from the machine and the blocking process started. At first, the containers intended for pouring paraffin were placed on the HOT PLATE device to heat the containers and bring them to the same temperature as the sample. Then, using a DISPENSER device, paraffin was poured into metal containers. The lid of the cassettes was opened and with the help of tweezers we put the sample in the container containing paraffin and the lid was closed. In this way, the blocks were prepared and ready to be cut. Sectioning of brain tissues containing black and white brain matter around the lateral ventricles was done according to the Paxinos and Watson atlas. The samples were cut using a microtome. After preparing the main cut, we put it in a hot water bath and removed it from the surface of the water using a slide. Then, the slide containing the section of the desired sample was placed in a special basket for staining. In the next step, we first put the slides in a 100-degree oven for 15 to 20 minutes to melt the excess paraffin and remove it from the sample. In the next step, we placed the samples in Xylenol to completely remove the excess paraffin from the samples (deparaffinization step). Then the samples were dried and placed in 70% alcohol for six minutes. In the next step, washing with distilled water was done, then they were placed in hematoxylin dye for 6 minutes. Then washing was done and in the next step, it was placed in eosin dye for 6 minutes. Then washing was done and then it was placed in 70% alcohol and then 90% alcohol thus the slides were prepared and microscopic images were taken with an optical microscope (Japan Olympus-BH2) and pathological markers were observed under the optical microscope.
ETHICAL PERMISSIONS
The method of this research was approved by the Medical Ethics Committee of Arak, Iran Azad University with code IR.IAU.ARAK.REC.1399.009.
STATISTICAL ANALYSIS
The analysis of the results was done based on the qualitative changes in the brain matters and the comparison of the sections of the experimental groups in terms of cell ischemia, vasogenic and hydrostatic parameters, and the typical microscopic slides were qualitatively compared. To compare the matter sections in terms of the amount and type of lesions, the scoring method was used by including zero (no damage) to four (maximum damage) and calculating the average damage in each group. Prism-GraphPad 9 software was used to analyze data and draw graphs. The comparison of groups was done with a one-way analysis of variance. Tukey's post-test was also used to determine the difference between each group. p value<0.05 was considered as the criterion of the significance level of difference between groups.
FINDING by TEXT
Microscopic examinations in the group receiving pure ethanol showed numerous pathological changes. Vasogenic edema, which could be seen with the strong expansion of the Virchow-Robin spaces as shown in Figure 1, this pathological change was caused by the effects of ethanol in the blood/brain barrier, and by increasing the intracranial pressure and reducing the blood supply to the neuronal cells, which are very sensitive to lack of oxygen and it leads to Cell damage changes. The difference between experimental groups in terms of vasogenic edema was significant (Figure 1); So the ethanol group showed the most damage compared to the negative control group (p<0.0001), while probiotic consumption reduced this damage compared to the ethanol group (p<0.01). Hydrostatic edema, which occurred due to the inflammation of nerve tissue and the blockage of the flow of cerebrospinal fluid in the cerebral ventricles, was visible as edema in the periphery of the cerebral ventricles (Figure 1). Also, the statistical comparison showed a significant difference between the ethanol group and the negative control group in terms of this injury (p<0.0001). Probiotic was able to reduce the significant difference in the damage caused by ethanol with the negative control group (p<0.0001). The results of this statistical comparison are shown in chart 2. Central chromatolysis in brain neurons was visible in the form of cell swelling, moving the nucleus to the cell periphery, and scattering of Nissl bodies (Figure 1). Neuronal ischemic damage was caused by blood supply disruption and caused a significant difference in this respect between the ethanol group and the negative control group (p<0.0001), although the use of probiotics along with ethanol could not compensate for this damage compared to the negative control group. But it caused a significant decrease in this regard compared to the ethanol group (p<0.01) (Chart 3). In these pathological changes, the neuronal cells were shriveled in a triangular shape with pyknosis nuclei that lost their central polarity in the cell cytoplasm (Figures 1 and 2). Following the spread of nerve tissue damage, the accumulation of glial cells was another pathological finding that indicated the extreme damage of nerve tissue under the influence of pure ethanol (Figure 3). In the group receiving ethanol along with probiotics, the changes were reduced by 15-25% compared to the group receiving pure ethanol, and the healthy groups and only probiotics had a healthy tissue pattern (Figure 4).
MAIN COMPARISON to the SIMILAR STUDIES
The healing effects of the probiotic on brain tissue are likely due to its ability to protect against ethanol-induced cellular damage and oxidative stress [13]. The difference between the ethanol and probiotic-ethanol groups may be due to the changes that ethanol has made in the brain tissue, especially around the lateral ventricles. Ethanol has led to cell damage by increasing intracranial pressure and reducing blood flow to hypoxia-sensitive nerve cells. Ischemic neuron damage is caused by blood flow disorder. The formation of significant amounts of brain acetaldehyde in vivo after ethanol consumption and its mechanism is not clearly defined, although catalase is a promising candidate [14]. Dendritic and synaptic changes have been documented in alcoholics, which receptor and transmitter changes, may explain the functional changes and cognitive deficits that result from more strong structural changes [15]. Based on the review of valid databases, this is probably the first study that investigated the effect of probiotics on the above tissue parameters in the acute ethanol administration model and based on the authors' review, there was no similar research to compare the results. But it is possible to analyze the effect of probiotics on the side effects of ethanol that indirectly lead to tissue changes in the brain. ... [16]. Probiotics reduce oxidative stress in alcohol-fed rodents. There is a possibility that this may be due in part to reduced CYP2E1 expression, another study showed that this finding may be the result of the healing of intestinal dysbiosis caused by alcohol (and oxidative stress), as it has been shown the case for probiotics and LactobacillusGG [17]. ... [18-24].
SUGGESTIONS
It is suggested to investigate the effect of nutritional supplements of other valuable probiotic strains on brain damage caused by ethanol. Also, the effect of the probiotic and prebiotic combination on brain poisoning caused by ethanol and other models of brain poisoning are studied.
LIMITATIONS
The limitations of the current research were mainly related to the unavailability of the studied probiotics. Also, due to the time limitation in the use of laboratory facilities, only the model of acute poisoning caused by ethanol was investigated.
CONCLUSIONS
Acute ethanol injection can cause visible pathological changes in the brain tissue, including vasogenic edema with strong expansion of the Virchow-Robin space, hydrostatic edema caused by (swelling or inflammation?) of nerve tissue, and (obstruction or blockage?) of cerebrospinal fluid flow in cerebral ventricles, central chromatolysis and ischemic neuronal damage. It follows disruption in blood supply and the accumulation of glial cells. A probiotic supplement can compensate for the tissue damage caused by ethanol in cortical and subcortical areas adjacent to cerebral ventricles. It appears that probiotics can exert their preventive pathways against alcohol toxicity through biochemical and microflora changes that improve brain tissue.
CLINICAL & PRACTICAL TIPS in POLICE MEDICINE
The use of ethanol causes damage to the white and gray matter of the brain and it is necessary to apply controls against its abuse. The consumption of probiotic food supplements in people under observation or control who suffered from ethanol abuse for any reason can have a compensatory and reducing effect on the damage of white and gray matter in the brain caused by it.
ACKNOWLEDGMENT
The researchers of this study thank TAKGENE Bio Company for providing probiotics in this research.
CONFLICT of INTEREST
The authors of the article state that there is no conflict of interest.
FUNDING SOURCES
This study was supported as a doctoral thesis by Arak, Iran Islamic Azad University.
Tables & Figures

Figure 1) Brain tissue of the healthy control group in which
the cells of the nerve tissue have a healthy pattern.
Hematoxylin/eosin staining and 40% magnification.

Figure 2) Ischemic cells undergo necrosis. Vasogenic edema is evident and indicated by the arrow (magnification 40%).
Severe hydrostatic swelling is visible at the margins of the brain ventricles (magnification 20%).

Figure 3) In the ethanol-probiotic group, the changes decreased by about 20% compared to the ethanol group

Figure 4) Microscopic view of brain tissue of ethanol-probiotic group.
Hematoxylin/eosin staining and 40% magnification.
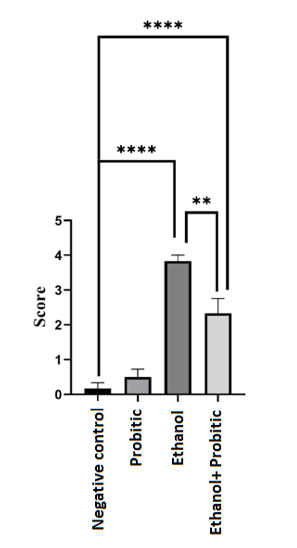
Chart 1) Comparison between experimental groups in terms of vasogenic with each other.
The ethanol group showed the highest damage compared to the negative control group (P<0.0001),
probiotic consumption did not reduce this difference but reduced this damage compared to
the ethanol group (P<0.01). Score shows the amount of waste observed in each sample.

Chart 2)- Statistical comparison of hydrostatic edema between experimental groups.
The ethanol group showed a significant difference in terms of this damage compared to the negative control group (P<0.0001).
Probiotics were able to reduce this significant difference in ethanol-induced damage with some negative control group (P<0.001).
Score shows the amount of waste observed in each sample.

Chart 3) Comparison of cellular ischemia between different experimental groups.
Ethanol use caused a significant difference in terms of this damage between the
ethanol and negative control groups (P<0.0001). But ethanol group had a significant
decrease in this regard (P<0.01). The score indicates the amount of waste observed in each sample.
Article Type: Original Research |
Subject:
Addiction & Substance Abuse
Received: 2022/01/24 | Accepted: 2022/06/6 | Published: 2022/07/1
Received: 2022/01/24 | Accepted: 2022/06/6 | Published: 2022/07/1
References
1. Airaodion AI, Ngwogu AC, Ekenjoku JA, Ngwogu KO. Hepatoprotective potency of ethanolic extract of Garcinia kola (Heckel) seed against acute ethanol-induced oxidative stress in Wistar rats. Int Res J Gastroenterol Hepatol. 2020;3(2):1-10. https://www.researchgate.net/publication/339051789_Hepatoprotective_Potency_of_Ethanolic_Extract_of_Garcinia_kola_Heckel_Seed_against_Acute_Ethanol-Induced_Oxidative_Stress_in_Wistar_Rats
2. Savage P, Horlock C, Stott B, Stebbing J. Enhanced sensitivity of lymphoid cells to ethanol ADH acetaldehyde toxicity; Implications for GDEPT and adoptive T cell therapy. CPPM. 2018;16(2):118-23. [DOI:10.2174/1875692116666180703151538]
3. Wang R, Zeng X, Liu B, Yi R, Zhou X, Mu J, Zhao X. Prophylactic effect of Lactobacillus plantarum KSFY06 on HCl/ethanol-induced gastric injury in mice. Food funct. 2020;11(3):2679-92. https://pubs.rsc.org/en/content/articlelanding/2020/fo/c9fo02474c [DOI:10.1039/C9FO02474C] [PMID]
4. Heidarzadeh F, Shahrabadi M, Rostami S, Pazoki M, Abbasi H, Hosseiniara SM, Hosseiniara S. Rapid and Accurate Diagnosis of Substance Abuse: A Narrative Review. J Police Med. 2022;11(1). http://dx.doi.org/10.30505/11.1.7
5. Dukay B, Walter FR, Vigh JP, Barabási B, Hajdu P, Balassa T, Migh E, Kincses A, Hoyk Z, Szögi T, Borbély E. Neuroinflammatory processes are augmented in mice overexpressing human heat-shock protein B1 following ethanol-induced brain injury. J Neuroinflammation. 2021(1):1-24. [DOI:10.1186/s12974-020-02070-2] [PMID] [PMCID]
6. Tizabi Y, Getachew B, Collins MA. Ethanol neurotoxicity. Handbook Neurotox. 2021:1-23. [DOI:10.1007/978-3-030-71519-9_205-1] [PMID] [PMCID]
7. Dudek I, Hajduga D, Sieńko C, Maani A, Sitarz E, Sitarz Met al. Alcohol-Induced Neuropathy in Chronic Alcoholism: Causes, Pathophysiology, Diagnosis, and Treatment Options. Curr Pathobiol Rep. 2020:87-97. [DOI:10.1007/s40139-020-00214-w]
8. Hammoud N, Jimenez-Shahed J. Chronic neurologic effects of alcohol. Clin Liver Dis. 2019;23(1):141-55. [DOI:10.1016/j.cld.2018.09.010] [PMID]
9. Westfall S, Lomis N, Kahouli I, Dia SY, Singh SP, Prakash S. Microbiome, probiotics and neurodegenerative diseases: deciphering the gut brain axis. Cell Mol Life Sci.. 2017;74(20):3769-87. [DOI:10.1007/s00018-017-2550-9] [PMID]
10. Zavareh AH, Khani RH, Pakpour B, Soheili M, Salami M. Probiotic treatment differentially affects the behavioral and electrophysiological aspects in ethanol exposed animals. Iran J Basic Med Sci. 2020;23(6):776. [DOI:10.22038%2Fijbms.2020.41685.9846]
11. Hadidi-Zavareh AH, Hajikhani R, Pakpour B, Salami M. Effect of probiotic supplementation on behavioral and electrophysiological aspects of memory and learning in ethanol exposed animals. KAUMS (FEYZ). 2020;24(1):1-9. [Persian]. http://feyz.kaums.ac.ir/browse.php?a_id=3909&sid=1&slc_lang=en
12. Parhizgar N, Azadyekta M, Zabihi R. Effect of probiotic supplementation on depression and anxiety. Complement Med J. 2021;11(2):166-79. [Persian]. http://cmja.arakmu.ac.ir/browse.php?a_id=807&sid=1&slc_lang=en&html=1 [DOI:10.32598/cmja.11.2.1073.1]
13. Warner DR, Warner JB, Hardesty JE, Song YL, King TN, Kang JX, Chen CY, Xie S, Yuan F, Prodhan MA, Ma X. Decreased ω-6: ω-3 PUFA ratio attenuates ethanol-induced alterations in intestinal homeostasis, microbiota, and liver injury. J lipid Res. 2019;60(12):2034-49. http://dx.doi.org/10.1194/jlr.RA119000200 [DOI:10.1194/jlr.RA119000200] [PMID] [PMCID]
14. Xu G, Li C, Parsiola AL, Li J, McCarter KD, Shi Ret al. Dose-dependent influences of ethanol on ischemic stroke: role of inflammation. Front cell Neurosci. 2019;13:6.
https://doi.org/10.3389/fncel.2019.00006 [DOI:10.3389%2Ffncel.2019.00006] [PMID] [PMCID]
15. Abrahao KP, Salinas AG, Lovinger DM. Alcohol and the brain: neuronal molecular targets, synapses, and circuits. Neuron. 2017;96(6):1223-38. [DOI:10.1016/j.neuron.2017.10.032] [PMID] [PMCID]
16. El-Sayyad HI. Future impact of probiotic therapy of metabolic disorder. J. Pharma. Rep. 2017;2:133. https://www.longdom.org/open-access/future-impact-of-probiotic-therapy-of-metabolic-disorder.pdf
17. Forsyth CB, Voigt RM, Keshavarzian A. Intestinal CYP2E1: A mediator of alcohol-induced gut leakiness. Redox Biol. 2014;3:40-6. [DOI:10.1016/j.redox.2014.10.002] [PMID] [PMCID]
18. Li YT, Xu H, Ye JZ, Wu WR, Shi D, Fang DQet al. Efficacy of Lactobacillus rhamnosus GG in treatment of acute pediatric diarrhea: A systematic review with meta-analysis. World J gastroenterol. 2019;25(33):4999.
https://doi.org/10.3748/wjg.v25.i33.4999 [DOI:10.3748%2Fwjg.v25.i33.4999] [PMID] [PMCID]
19. Tsai YS, Lin SW, Chen YL, Chen CC. Effect of probiotics Lactobacillus paracasei GKS6, L. Plantarum GKM3, and L. rhamnosus GKLC1 on alleviating alcohol-induced alcoholic liver disease in a mouse model. Nutrition Res Pract. 2020;14(4):299-308.
https://doi.org/10.4162/nrp.2020.14.4.299 [DOI:10.4162%2Fnrp.2020.14.4.299] [PMID] [PMCID]
20. Calasso M, Di Cagno R, De Angelis M, Campanella D, Minervini F, Gobbetti M. Effects of the peptide pheromone plantaricin A and cocultivation with Lactobacillus sanfranciscensis DPPMA174 on the exoproteome and the adhesion capacity of Lactobacillus Plantarum DC400. Appl Environ. Microbiol. 2013;79(8):2657-69.
https://doi.org/10.1128/AEM.03625-12 [DOI:10.1128%2FAEM.03625-12] [PMID] [PMCID]
21. Leclercq S, Stärkel P, Delzenne NM, de Timary P. The gut microbiota: a new target in the management of alcohol dependence? Alcohol. 2019;74:105-11. [DOI:10.1016/j.alcohol.2018.03.005] [PMID]
22. Lee E, Lee JE. Impact of drinking alcohol on gut microbiota: Recent perspectives on ethanol and alcoholic beverage. Curr. Opin Food Sci. 2021;37:91-7. [DOI:10.1016/j.cofs.2020.10.001]
23. Abdelhamid M, Zhou C, Ohno K, Kuhara T, Taslima F, Abdullah M et al. Michikawa M. Probiotic Bifidobacterium breve Prevents Memory Impairment Through the Reduction of Both Amyloid-β Production and Microglia Activation in APP Knock-In Mouse. J Alz Dis. 2021;1-7.
https://doi.org/10.3233/JAD-215025 [DOI:10.3233/jad-215025] [PMID] [PMCID]
24. La Fata G, Weber P, Mohajeri MH. Probiotics and the gut immune system: indirect regulation. Probiotics Antimicrob Proteins. 2018;10(1):11-21. [DOI:10.1007/s12602-017-9322-6] [PMID] [PMCID]
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |

